Proven Medical Device Contract Manufacturing Services
Our leading-edge equipment, materials expertise, and long-term customer relationships have helped leading medical and dental OEMs pioneer new advancements in care.
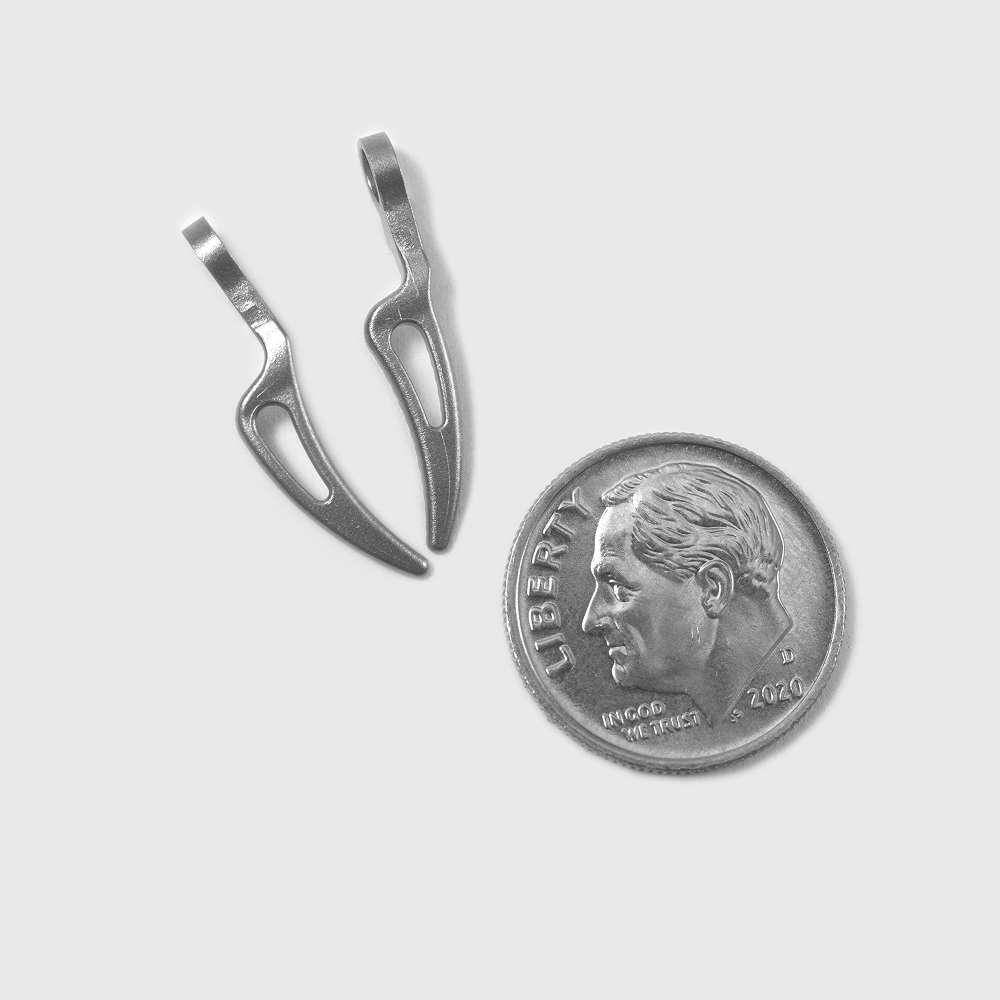
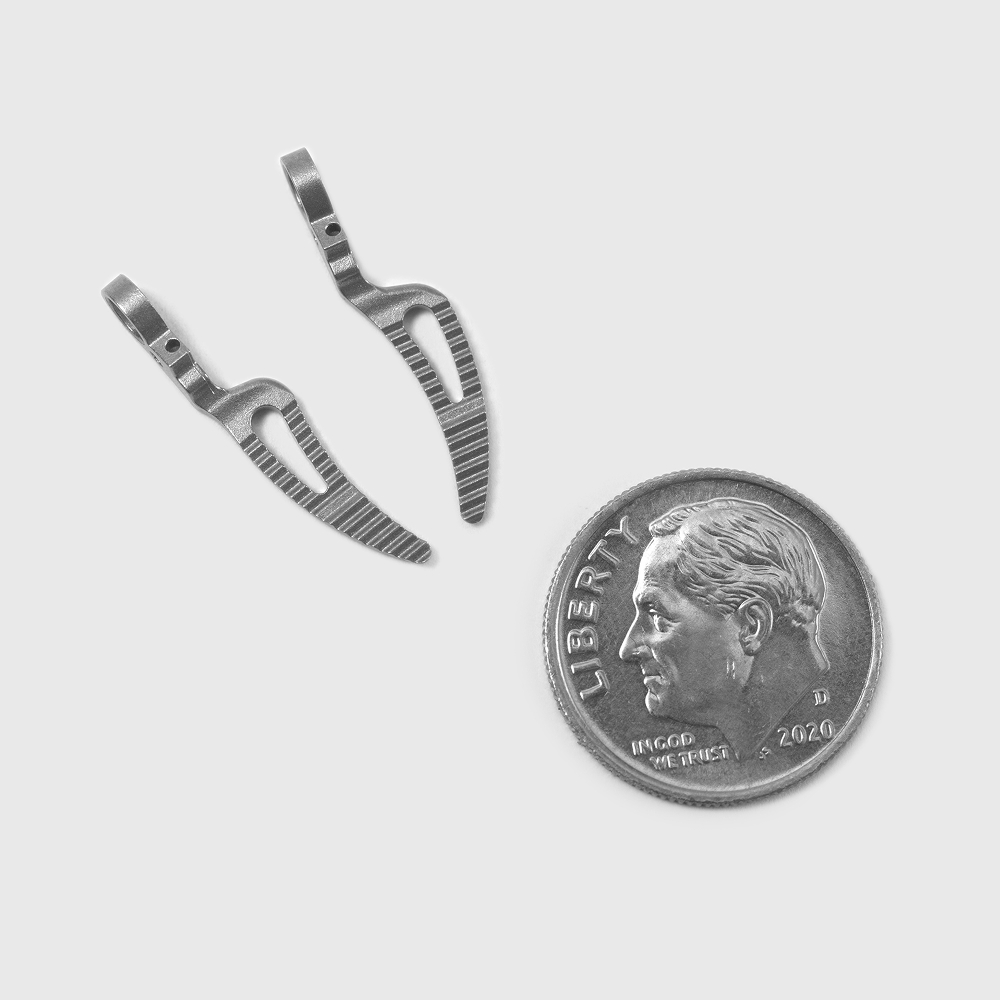
Designed for Critical Medical Applications
Whether you’re designing instruments, implants, or diagnostic devices, our advanced molding, cutting, and stamping capabilities support a wide range of products for the most demanding specifications:
- Minimally invasive surgical equipment
- Test equipment parts
- Surgical stapling components
- Orthopedic fixation components
- Drug-delivery device components
- Diagnostic and test equipment components
- And more
The Power of Vertical Integration
Mitigate risk and reduce unknowns by working with a single partner, from prototype to production. Our full-scale capabilities and expert guidance facilitate tighter control over production, timelines, and budget.
Metal & Ceramic Injection Molding (MIM/CIM)
The ease of injection molding meets the strength and functionality of machined metals, for virtually any design, regardless of complexity.
Metal 3D Printing: PureForm
Complex, precision metal parts are produced via PureForm™, Greene Group Industries’ dedicated production-ready metal printing technology.
Precision CNC Machining
60+ CNC mills, lathes, wire EDM machines, multi-axis mills, and turn centers support extremely complex designs with unmatched quality, from prototype to production.
Metal Stamping
State-of-the-art die-making capabilities that maintain precise stampings of 100+ major and minor dimensions.
Plastic Overmolding
Ultra-precise mechanical and polymer integration that preserves dimensional stability and adhesion.
Automated Assembly
In-house automated assembly minimizes the risk of failure between parts and performance.
Mold Design & Manufacture
Expert-led holistic approach to mold design with production in mind.
Frequently Asked Questions
We understand that OEMs in the medical field face unique regulatory and performance challenges, and we have the processes and capabilities in place to meet them.
What quality controls are in place at Greene Group Industries?
We maintain a rigorous, self-imposed “total quality system” to ensure efficiency, facility cleanliness, and product performance. In addition, we are AS9100-compliant and certified to ISO 9001:2015 and ISO 13485 for medical device contract manufacturing. Our process emphasizes full traceability, process validation, and audit readiness.
Where does production take place?
Our manufacturing happens at U.S.-based facilities. For more information, see our Facilities page.
Can GGI support both early-stage development and long-term production scaling?
Yes. We routinely support OEMs from prototyping and pilot builds through validated, high-volume production. Our manufacturing and capacity planning approach is designed to accommodate design evolution early on while ensuring stable, repeatable output as programs scale.
What value-added services does GGI offer?
Our integrated approach means we also deliver state-of-the-art finishing processes, such as zinc phosphate, electroless nickel, and gold plating. We also provide heat treatment, welding, plastic overmolding, and automated production and assembly services. These end-to-end capabilities reduce risk for our clients by ensuring the same high level of quality and consistency at every stage.
Ready to Start Your Next Project?
See how our full-service contract manufacturing capabilities can elevate your precision component production and performance.